AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Carbon dioxide formula mass1/16/2024  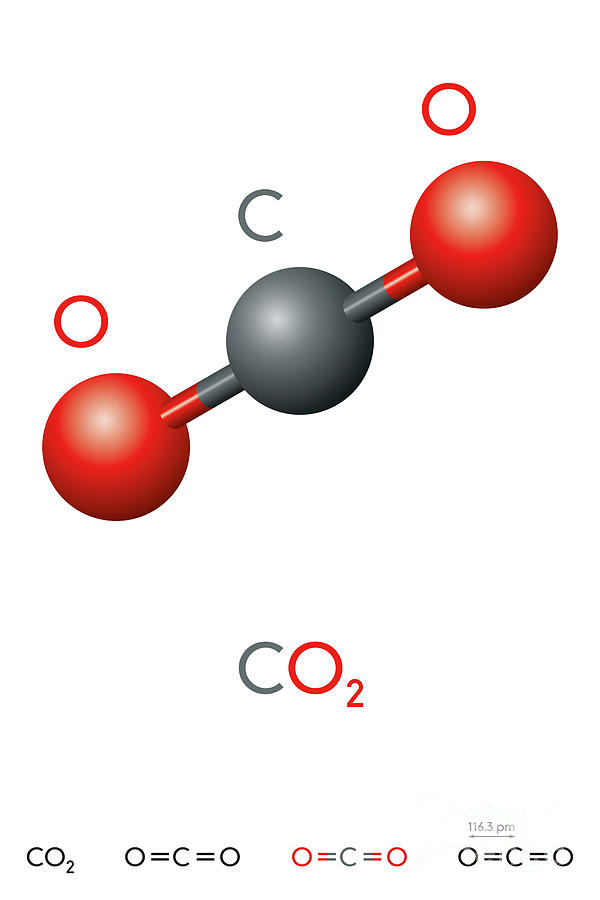
Subtract the mass of the glass flask in 7) from the mass of the flask, foil and carbon dioxide as measured in 4) above. Subtract the mass of dry air from the mass of the flask and foil as measured in 2) above.Ĩ) Calculate the mass of carbon dioxide in the flask. You will have noticed that all the dry ice has sublimed.Ĥ) Record the atmospheric pressure and temperature.Ĭonvert the atmospheric pressure into kilopascals and the temperature into Kelvin.ĥ) Measure the volume of the flask by measuring the volume of water it takes to fill it to the brim.Ħ) Calculate the mass of air in the flask at room temperature using the table of the density of dry air on the right.ħ) Calculate the mass of the glass flask. 
When the mass is constant for more than 20 seconds record the final reading. Figure 2.3.2: Italian scientist Amedeo Avogadro, whose work led to the concept of the mole as a counting unit in chemistry. carbon dioxide carbonic anhydride Dry ice 124-38-9 carbonic acid gas. There also would be 6.02 × 1023 bananas in a mole of bananas, if such a huge number of bananas ever existed. There are 6.02 × 1023 water molecules in a mole of water molecules. 
Record the mass of the flask and carbon dioxide. There are 6.02 × 1023 atoms in a mole of carbon. What is the average atomic mass of M Consider a gaseous binary compound with a molar mass of 62.09. Observe the mass dropping as carbon dioxide escapes. Equation 1 is a balanced equation one mole of methane reacts with two moles of dioxygen to produce one mole of carbon dioxide and two moles of water. The products of the reaction are carbon dioxide (C). 3) With a carbon dioxide cylinder create some dry ice.Ĥ) Place about 2.00 grams into the flask and place it on the electronic scale. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
